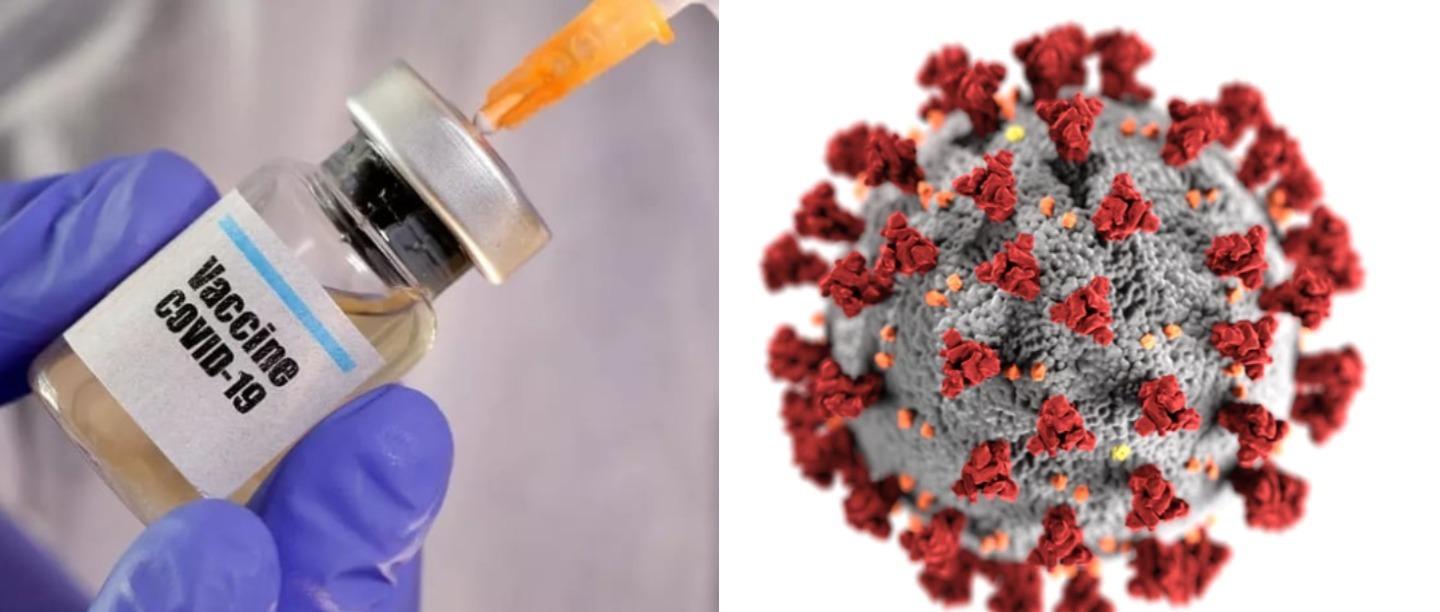
As scientists and major medical players across the world race to find a COVID-19 cure, the much talked about experimental vaccine developed by Oxford University and AstraZeneca (a global, science-led biopharmaceutical company) has brought a ray of hope. In a recently published study, it was stated that the initial trials of the vaccine suggest that it doesn’t lead to any major side effects and prompts double immunity in the subjects.
While the vaccine is yet to enter its phase 3 of its trials, the findings bear great hope especially in a scenario when COVID-19 cases continue spiking across the world with no foolproof cure at the moment. The findings from these trials are also crucial given that Serum Institute of India (SII), the largest vaccine manufacturer in the world, has been chosen by Oxford and AstraZeneca to manufacture the vaccine once it gets ready.
In a recent media interaction, SII’s chief executive officer, Adar Poonawalla shared that the large-scale trials of this vaccine will soon start in India. They have named it Covishield and it is the first COVID-19 vaccine that SII hopes to launch if the trials are successful both in India and the UK. Additionally, the company has also decided to start with the production, which is a huge commercial risk given that the vaccine is still in its trial phase.
Now, as SII invests great hope in the vaccine and projects to produce up to 3 to 4 million doses by December, this is indeed huge news given that India is already suffering from an inundating caseload. Think Oxford’s promising vaccine would finally break the COVID-19 cycle? Here’s everything that you need to know about the vaccine:
How Has It Been Developed?
Named ChAdOx1 nCoV-19 by the Oxford researchers, the vaccine is made by genetically engineering adenovirus (the same virus that causes the common cold in chimpanzees). The virus has been fused with the genetic material of the spike protein of the SARS-CoV-2. This spike protein is what gives the novel coronavirus its unique shape and single-handedly leads coronaviruses to enter host cells and latch on to the cells. The virus thus formed by these scientists has been heavily modified, firstly to ensure that it doesn’t cause infections in people and secondly to make it “look” more like coronavirus. The initial trials of the vaccine suggest that it is in fact potent enough to trigger double immunity in the subject.
How Does ChAdOx1 nCoV-19 Trigger Double Immunity?
The initial trials as published in Lancet suggest that the vaccine actually elicited antibody and T-cell immune responses (Cell-mediated immune responses involve the destruction of infected cells by cytotoxic T cells) in the subjects and that the strongest response was recorded in subjects who had been given double dosage of the vaccine.
Now, while most of focus all this while has been about producing antibodies to combat coronavirus, it is important to understand that almost all effective vaccines need more than that to be actually successful. This is where the T-cell response comes into play. A type of white blood cell, T cells help coordinate the immune system and also work by spotting and eliminating all those body cells that have been infected by the targeted virus.
The trial results for ChAdOx1 nCoV-19 show that levels of T-cells and antibody levels are peaking respectively 14 and 28 days after the vaccination.
Where Does It Stand In Turn Of The Trial Timeline?
Now that the initial trials have shown promising results, the next phase of trials will be conducted on a larger population. While the trials were to be administered on 10,000 people in the UK, they have no been expanded to other countries owing to lower caseload in the country. In India alone, approximately 5,000 Indian volunteers (mostly from Maharasthra) would be participating in the trials by the end of August.
How Soon Will India Get A Vaccine?
“We will be going for large scale manufacturing in mid-August-early-August… By the end of this year, we should be able to produce 3 to 4 million doses come the end of December. That’s the target and I hope we can do that,” Adar Poonawalla shared in a recent statement.
Given these projections and computing them with the time required to finish the trials and all the permissions to get cleared, Poonawalla hopes that the vaccines would be available by the first quarter of 2021.
Additionally, SII has also ensured that the vaccine would be available at very affordable prices in the country. “We are going to give it at a very affordable price… We are planning to put it at about Rs 1000 or less than that… I don’t think any citizen of India or of any other country is going to have to pay for it because it is going to be bought by the government and distributed free,” he added.
As SII invests great hope in the Oxford vaccine, so do we. Here’s hoping that it proves to be a success and is available for general use as soon as possible!
Featured Image: Instagram