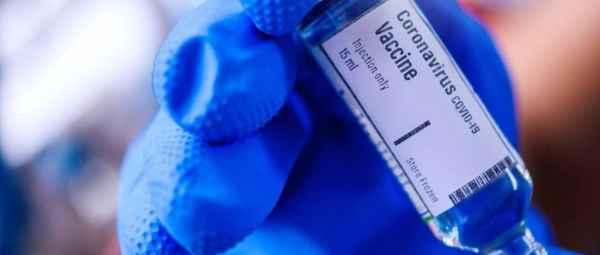
The coronavirus outbreak is possibly the worst pandemic most of us have witnessed in our lifetime, infecting 29,95,152 people so far and killing 2,07,008 of them. And while most countries across the world have imposed strict lockdowns to slow the spread of the virus, it is only a temporary solution. After all, we can’t stay locked up indoors forever, especially because it has severely affected the economy. While doctors are exploring multiple treatment options, including various drugs and therapies, a vaccine is our best bet to permanently defeat the virus.
The unfortunate news is that vaccines can’t just be developed overnight–they need to go through multiple levels of testing to ensure they are 100% safe for the masses. On average, a vaccine can either take a year or several years to finally hit the market. Thankfully, doctors and medical experts are working round the clock to deliver a vaccine to treat COVID-19.
Several companies have started working on vaccines, and clinical trials have already begun worldwide. The University of Oxford began testing its vaccines on human subjects on April 23, five other companies from across the globe have also reached the testing phase. Wondering how long the vaccine will take to reach India? Well, Pune-based firm Serum Institute of India (SII) said on Sunday that it will likely begin the production of a COVID-19 vaccine developed by Oxford University in the next two-three weeks.
The company has partnered with Oxford University as one of the seven global institutions manufacturing the vaccine, and hopes to bring it to the market by October if human clinical trials are successful.

Shutterstock
“SII will be manufacturing the vaccine in anticipation of clinical trials succeeding by September-October in the UK. Following that, SII has undertaken the decision to initiate the manufacture at their own risk. The decision has been solely taken to have a jump-start on manufacturing, to have enough doses available, if the clinical trials work,” said Adar Poonawalla, chief executive officer of the Indian vaccine major, said in a statement.
He added that the vaccines will be manufactured at the company’s facility in Pune as building a new facility for COVI-19 vaccines would have taken around two-three years.
The Oxford trials began last Thursday, and a total of 800 volunteers will be injected with ChAdOx1 nCoV-19 for the purpose of the study. If the trials are a success, scientists hope to have one million doses ready by September and to dramatically scale up manufacturing after that.
“Our team has been working closely with Dr Hill ( professor Adrian Hill) from Oxford University, and we are expecting to initiate production of the vaccine in two-three weeks and produce five million doses per month for the first 6 months, following which, we hope to scale up production to 10 million doses per month,” Poonawalla told news agency Press Trust of India.
Also Read: On The Path To A Coronavirus-Free World: India’s COVID-19 Cases Now Doubling In 7.5 Days
According to the researchers at Oxford University, the vaccine is made from a weakened version of a common cold adenovirus taken from chimps and genetically modified to make it impossible for it to infect humans. To develop the vaccine, researchers added genetic material to ChAdOx1 from the Sars-CoV-2 virus’s surface protein, spike glycoprotein (S), which helps the virus to bind to Ace2 receptors to enter human cells and cause an infection.
This is certainly a positive development in our collective fight against COVID-19, and we are grateful to medical professionals working towards this vaccine, as well as those in the hospitals risking their own lives to save ours. Let’s do our bit by following their advice–practice social distancing, wash your hands frequently and self-isolate as soon as you notice any symptoms.
Featured Image: Instagram